The Global Neutropenia Treatment Market size is expected to reach $18.9 billion by 2026, rising at a market growth of 6.5% CAGR during the forecast period. Neutropenia is an abnormal condition characterized by a critically low level of circulating neutrophils, a type of WBC found in the blood. These cells make up the majority of circulating lymphocytes and help protect the body from infections caused by bacteria, viruses, and other pathogenic organisms. A decrease in the amount of neutrophil in the blood allows a patient to be vulnerable to infection and can also prove fatal. The condition, therefore, requires immediate prophylaxis.
Global Neutropenia Treatment Market Size
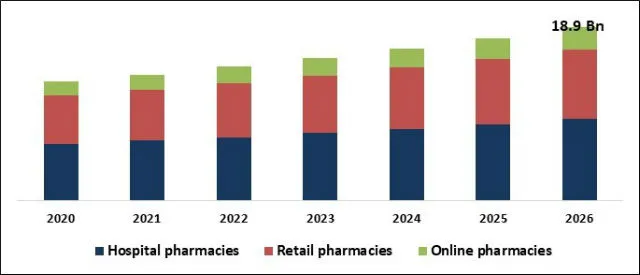
Novel treatments combined with growing cases of chemotherapy-induced neutropenia are one of the primary drivers of market growth. In addition, the novel drug delivery strategy also increases patient convenience, leading to greater adherence to the patient and thereby stimulates the growth of the market. Moreover, since cancer is a known cause of developing neutropenia, increasing cases of chemotherapy for cancer treatment will be a growth driver for the neutropenia treatment market.
Neutropenia Treatment Market Share
One of the new developments to gain momentum in the global neutropenia treatment market is the introduction of small molecules. The development of Plinabulin, a small molecule developed by BeyondSpring Pharmaceuticals with immune-boosting effects, is expected to revolutionize the therapeutic regimen for neutropenia. In addition to the immune-boosting effects, the Phase II studies have revealed much more beneficial anti-cancer effects of this molecule. Currently, there is a high demand for cost-effective drugs for treatment in the neutropenia treatment market. As a result, vendors are focused on the production of small molecules with lower processing costs than biologics.
Free Valuable Insights: Global Neutropenia Treatment Market to reach a market size of $18.9 billion by 2026
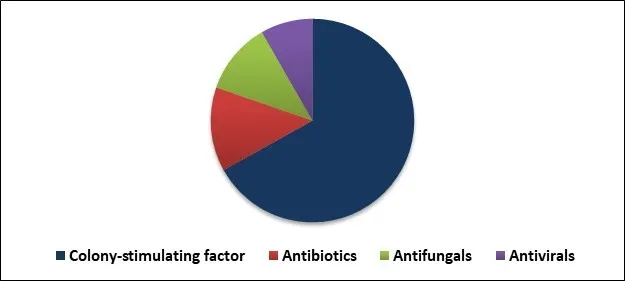
Based on Distribution channel, the market is segmented into Retail pharmacies, Hospital pharmacies and Online pharmacies. Based on Treatment, the market is segmented into Colony-stimulating factor, Antibiotics, Antifungals and Antivirals. Based on Regions, the market is segmented into North America, Europe, Asia Pacific, and Latin America, Middle East & Africa.
The market research report covers the analysis of key stake holders of the market. Key companies profiled in the report include Amgen, Inc., Novartis AG, Pfizer, Inc., Teva Pharmaceuticals Industries Ltd., BeyondSpring, Inc., Spectrum Pharmaceuticals, Inc., Kyowa Kirin Co., Ltd. (Kirin Company), Mylan N.V., Cellerant Therapeutics, Inc., and Partner Therapeutics, Inc.
Market Segmentation:
By Distribution Channel
By Treatment
By Geography
Companies Profiled

The neutropenia treatment market is projected to reach USD 18.9 billion by 2026.
The major factors that are anticipated to drive the neutropenia treatment industry include infections associated with chemotherapy-induced neutropenia, Strong drug pipeline, and A rise in the incidence of cancer cases.
The colony-stimulating factor market dominated the global neutropenia treatment market by treatment in 2019.
The neutropenia treatment market has witnessed a significant rise in North America and is expected to dominate the market over the forecast period.
mgen, Inc., Novartis AG, Pfizer, Inc., Teva Pharmaceuticals Industries Ltd., BeyondSpring, Inc., Spectrum Pharmaceuticals, Inc., Kyowa Kirin Co., Ltd. (Kirin Company), Mylan N.V., Cellerant Therapeutics, Inc., and Partner Therapeutics, Inc.
Our team of dedicated experts can provide you with attractive expansion opportunities for your business.

