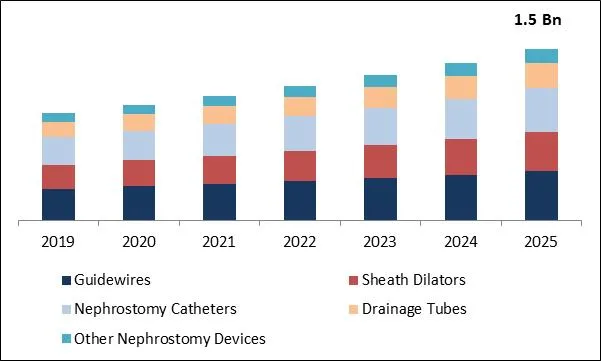
The Global Nephrostomy Devices Market size is expected to reach $1.5 billion by 2025, rising at a market growth of 8% CAGR during the forecast period. The growing popularity of percutaneous nephrostomy processes is a key factor in creating potential growth opportunities for the nephrostomy device industry. Several complications are required for traditional open-label operations, which lead to increased demands for minimally invasive operations like the PCN. For the blockage in the upper urinary tract, PCN is a minimally invasive procedure. This procedure is used as a first-line treatment for patients with kidney stones larger than 2 cm. The incision is about 1 cm lower and results in faster recovery and shorter hospitalization stays.
Global Nephrostomy Devices Market Size
Nephrostomy is an opening that exists between the skin and the kidney. A nephrostomy tube that is passed from the back, is a thin plastic tube inserted through the skin, into the kidney, to the spot where urine is collected. The task of the tube is to drain the blocked urine. This enables the proper functioning of the kidney and protects it from further damage. Nephrostomy tubes help to clear out any infection. The entire procedure is guided by ultrasound and/or X-ray images (or pictures). Contrast has also injected the part of the kidney which collects urine. With this, the tube can easily drain the kidney to be inserted into the collecting system.
Nephrostomy catheter placement is mostly conducted by interventional radiologists through image guidance within radiology or operating room. The most prevalent cause behind nephrostomy is the cancer of the reproductive organs. Dilation catheters are major preferences for treating percutaneous dilation challenges. Though contraindicated for pregnant women, percutaneous nephrostomy is suggested for certain patients who show symptoms of urolithiasis during pregnancy. Nephrostomy devices are essential in cases where patients suffer from urinary tract infections, benign prostate hyperplasia, bladder prolapse, urinary incontinence, etc.
Global Nephrostomy Devices Market Share
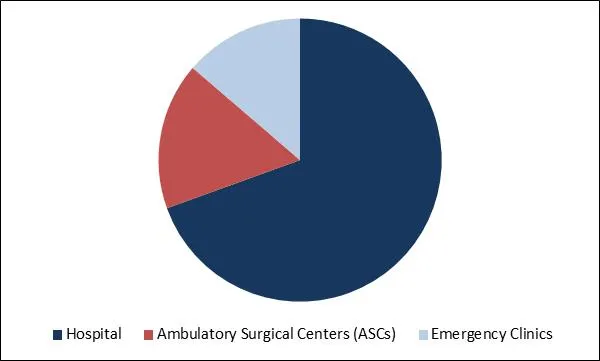
Based on Product, the market is segmented into Guidewires, Sheath Dilators, Nephrostomy Catheters, Drainage Tubes and Other Nephrostomy Devices. Guidewire manufacturers focus on working with healthcare professionals to increase their base of customers and to spike their industry presence. Thus the demand for guidewire is expected to grow with the manufacturers' initiatives and the presence of well-established and skilled workers in developed economies. Technological advances in nephrostomy devices are projected to offer the market profitable opportunities like introducing the Balloon Dilation Catheter with original multi-layer technology. Based on End Use, the market is segmented into Hospital, Ambulatory Surgical Centers (ASCs) and Emergency Clinics. Based on Regions, the market is segmented into North America, Europe, Asia Pacific, and Latin America, Middle East & Africa.
Nephrostomy Devices Market Cardinal Matrix
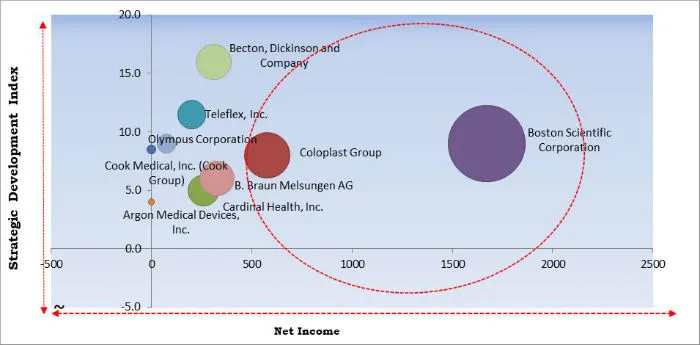
The major strategies followed by the market participants are Product launches and Partnerships & Collaborations. Based on the Analysis presented in the Cardinal matrix, Coloplast Group and Boston Scientific Corporation are some of the forerunners in the Nephrostomy Devices Market. Companies such as Becton, Dickinson and Company, Teleflex, Inc., Olympus Corporation, Cook Medical, Inc. (Cook Group), B. Braun Melsungen AG, Cardinal Health, Inc., and Argon Medical Devices, Inc. are some of the key innovators in Nephrostomy Devices Market. The market research report covers the analysis of key stake holders of the market. Key companies profiled in the report include Boston Scientific Corporation, Teleflex, Inc., Cook Medical, Inc. (Cook Group), Coloplast Group, Olympus Corporation, Argon Medical Devices, Inc., UreSil LLC, B. Braun Melsungen AG, Cardinal Health, Inc., and Becton, Dickinson and Company.
These companies focus more on new product development, technological advancements, and M&A to strengthen their foothold in the global market. For Instance, In May 2019, Cordis, a Cardiac Health company launched RADIAL 360 portfolio which has RAIN Sheath Transradial Thin-Walled Introducer, RBL-TG and RBL-JK Universal Shapes for the INFINITI and SUPER TORQUE PLUS Diagnostic Catheters, RAILWAY Sheathless Access System, etc. for the improvement in patient comfort. Coloplast took over IncoCare (Denmark) for the expansion of its product portfolio in February 2018.
» Product Launches:
» Acquisitions:
» Partnerships, Collaborations and Agreements:
Market Segmentation:
By Product
By End Use
By Geography
Companies Profiled

Our team of dedicated experts can provide you with attractive expansion opportunities for your business.

