The Global Meningococcal Vaccine Market size is expected to reach $4.5 billion by 2026, rising at a market growth of 8.5% CAGR during the forecast period. Meningococcal meningitis is a bacterial infection that is caused by gram-negative bacteria, Neisseria meningitides that includes a spectrum of infections such as bacteremia (meningococcemia), meningitis, and bacteremia pneumonia.
The meningococcal disease is very rare but is highly fatal, if left untreated, up to 50% of the patients die or get some serious complications thereafter. In meningococcal meningitis, bacteria infect the lining of the brain and spinal cord and can potentially cause large endemics as well as epidemics. The disease is transmitted from an infected person through direct contact with droplets of the throat or respiratory secretions, asymptomatic carriers can also capable of transmitting infection.
There is an increasing prevalence of various types of bacteria that cause meningococcal meningitis. The most common type of meningitis causing bacteria are A, B, C, W, Y, and type X that was more lately reported in Africa, continue to underline the criticality of more effective vaccine of meningococcal to be available in the coming years.
Over the past few decades, cases of meningitis have been increased and are one of the key factors likely to drive the market for meningococcal vaccines. According to the Centers for Disease Control and Prevention (CDC), about 1.2 million cases of meningococcal meningitis are estimated to occur worldwide every year. The government and regulatory authorities are taking initiatives to combat the increasing prevalence of meningitis are likely to drive the market growth. For example, “Defeating meningitis by 2030” was introduced in 2018 at the World Health Assembly by Eastern Mediterranean and African regions. This program is focused to develop a strategic roadmap for the prevention of meningitis by prioritizing research and improved control activities.
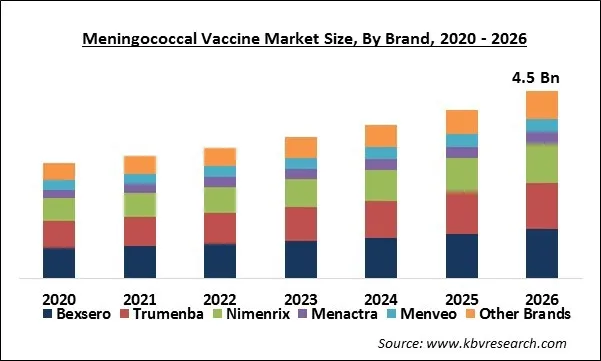
Based on Brand, the market is segmented into Bexsero, Trumenba, Nimenrix, Menactra, Menveo and Other Brands. The MenACWY vaccine and MenC vaccine are expected to experience swift growth during the forecast period. This is accredited to the increased adoption of these vaccines in several countries across the globe through immunization programs. Furthermore, outbreaks of meningococcal in several countries resulted in an increasing demand for meningococcal vaccines, which further boost the growth of the meningococcal vaccine market.
Based on Type, the market is segmented into Bivalent, Quadrivalent and Other Types. Quadrivalent meningococcal vaccines had the largest revenue share of the market in 2019, owing to the increased used brands like Menveo, Menactra, and Nimenrix, for this category. New vaccines are increasingly approved that will protect people against meningococcal meningitis and is expected to boost the growth of this market.
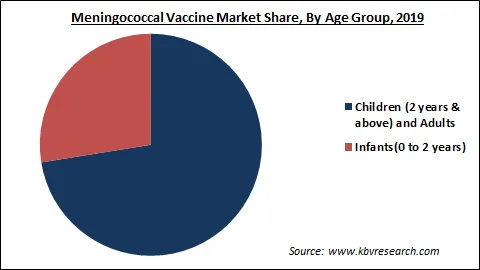
Based on Age Group, the market is segmented into Infants (0 to 2 years) and Children (2 years & above). The infant segment is anticipated to be the fastest-growing segment in the coming years due to the increasing incidence of meningitis among infants. According to the European Centre for Disease Prevention and Control (ECDC), IMD incidence was observed highest in infants in 2017. In Europe, there were nearly 8.2 confirmed cases per 100,000 populations in children below one year.
| Report Attribute | Details |
|---|---|
| Market size value in 2019 | USD 2.6 Billion |
| Market size forecast in 2026 | USD 4.5 Billion |
| Base Year | 2019 |
| Historical Period | 2016 to 2018 |
| Forecast Period | 2020 to 2026 |
| Revenue Growth Rate | CAGR of 8.5% from 2020 to 2026 |
| Number of Pages | 201 |
| Number of Tables | 370 |
| Report coverage | Market Trends, Revenue Estimation and Forecast, Segmentation Analysis, Regional and Country Breakdown, Companies Strategic Developments, Company Profiling, Competition Analysis |
| Segments covered | Brand, Type, Age Group, Region |
| Country scope | US, Canada, Mexico, Germany, UK, France, Russia, Spain, Italy, China, Japan, India, South Korea, Singapore, Malaysia, Brazil, Argentina, UAE, Saudi Arabia, South Africa, Nigeria |
| Growth Drivers |
|
| Restraints |
|
Based on Regions, the market is segmented into North America, Europe, Asia Pacific, and Latin America, Middle East & Africa. North America led the market and had a significant revenue share of the market in 2019 which is followed by Europe. Favorable approvals, supportive initiatives by the government, and high investments in R&D are the major factors fueling the North American market.
Free Valuable Insights: Global Meningococcal Vaccine Market to reach a market size of $4.5 Billion by 2026
The market research report covers the analysis of key stake holders of the market. Key companies profiled in the report include GlaxoSmithKline PLC (GSK), Pfizer, Inc., Sanofi S.A., Walvax Biotechnology Co., Ltd., Bio-Manguinhos (Oswaldo Cruz Foundation), Bio-Med Pvt. Ltd., Chongqing Zhifei Biological Products Co., Ltd., Hualan Biological Engineering, Inc., Incepta Pharmaceuticals Ltd. and Serum Institute of India Pvt. Ltd. (Poonawalla Investments & Industries Pvt. Ltd.).
By Brand
By Type
By Age Group
By Geography
Companies Profiled

The global meningococcal vaccine market size is expected to reach $4.5 billion by 2026.
There are several reason that cause high demand of this market one of them is increase in the outbreak of meningococcal disease.
The Quadrivalent market dominated the Global Meningococcal Vaccine Market by Type in 2019, growing at a CAGR of 8.2 % during the forecast period.
GlaxoSmithKline PLC (GSK), Pfizer, Inc., Sanofi S.A., Walvax Biotechnology Co., Ltd., Bio-Manguinhos (Oswaldo Cruz Foundation), Bio-Med Pvt. Ltd., Chongqing Zhifei Biological Products Co., Ltd., Hualan Biological Engineering, Inc., Incepta Pharmaceuticals Ltd. and Serum Institute of India Pvt. Ltd. (Poonawalla Investments & Industries Pvt. Ltd.).
Our team of dedicated experts can provide you with attractive expansion opportunities for your business.

