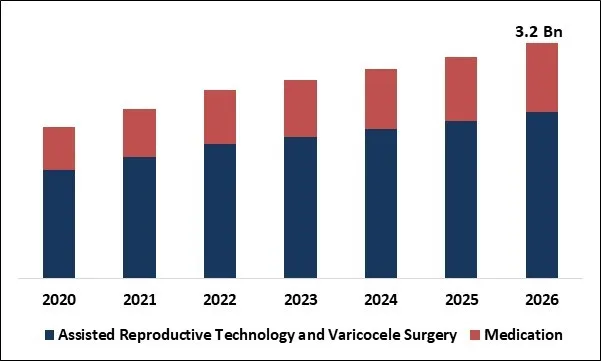
The Global Male Infertility Market size is expected to reach $3.2 billion by 2026, rising at a market growth of 7.6% CAGR during the forecast period. Male infertility is the inability of a male to cause pregnancy in a fertile female. A male's fertility relies mainly on the quantity and quality of their sperms. If a man's sperm count is small or if the sperm is of poor quality, it would be difficult, and even impossible, for him to cause pregnancy. The problem of infertility is widespread. Male fertility includes the normal functioning of the hypothalamus, pituitary gland, and testes. Nonetheless, 40 percent-50 percent of cases have no identifiable cause and 30 percent-40 percent of cases are due to problems in the testes.
Global Male Infertility Market Size
Continuous R&D efforts are being made to better understand the idiopathic world of infertility. As a result, companies are investing in the development of root cause identification and targeted treatment devices. In addition, the production and commercialization of hormonal therapy are expected to fuel the demand for male infertility during the forecast period. Governments in different countries have taken steps to boost the recovery scenario for infertility-related therapies. In addition, improved safety of patient data due to standardization of legislation is expected to increase consumer demand for infertility treatment.
Advancements in medical testing and rising infertility in males are two of the main factors driving the demand for male infertility. In addition, lifestyle-associated disorders causing infertility, rising obesity, and the expanding aging population are likely to boost the market growth over the forecast period. In the U.S., one-third of cases of infertility are attributed to female factors, one third to male causes, and the other one third is uncertain.
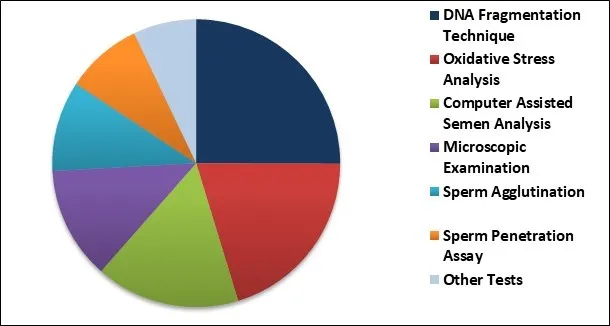
Male Infertility Market Share
Free Valuable Insights: Global Male Infertility Market to reach a market size of $3.2 billion by 2026
ARTs are used to cure major factors responsible for male infertility. These factors range from sperm autoantibodies, viral orchitis, epididymal dysfunction, accessory gland infections, chemotherapy, to heat, trauma, and other idiopathic factors. High treatment costs and the requirement for several other treatment cycles are some of the key factors contributing to the growth of the segment. Growing spending on health care in developed countries, raising awareness of male infertility, and growing acceptance in middle and low-income countries are the main factors projected to drive the ART segment over the forecast period.
Based on Treatment, the market is segmented into Assisted Reproductive Technology & Varicocele Surgery and Medication. Based on Test, the market is segmented into DNA Fragmentation Technique, Oxidative Stress Analysis, Computer Assisted Semen Analysis, Microscopic Examination, Sperm Agglutination, Sperm Penetration Assay and Other Tests. Based on Regions, the market is segmented into North America, Europe, Asia Pacific, and Latin America, Middle East & Africa.
The market research report covers the analysis of key stake holders of the market. Key companies profiled in the report include Sanofi S.A., Bayer AG, Cadila Healthcare Ltd. (Zydus Cadila), Endo International PLC, Aytu BioScience, Inc., Vitrolife AB, SCSA Diagnostics, Inc., Andrology Solutions Limited, Halotech DNA SL, and Intas Pharmaceutical Ltd.
Market Segmentation:
By Treatment
By Tests
By Geography
Companies Profiled

According to a new report published by KBV Research, The male infertility market is projected to reach USD 3.2 billion by 2026.
The major factors that are anticipated to drive the male infertility industry include increasing incidence of infertility, growing demand for assisted reproductive technologies [ART], and changing lifestyle, increasing ages, excess consumption of alcohol and drugs, environmental factors and genetic abnormalities.
The North America dominated the regional segment of the male infertility market and held the largest share of total revenue in 2019.
Sanofi S.A., Bayer AG, Cadila Healthcare Ltd. (Zydus Cadila), Endo International PLC, Aytu BioScience, Inc., Vitrolife AB, SCSA Diagnostics, Inc., Andrology Solutions Limited, Halotech DNA SL, and Intas Pharmaceutical Ltd.
Our team of dedicated experts can provide you with attractive expansion opportunities for your business.

