“Global Laboratory Developed Tests Market to reach a market value of USD 20.3 Billion by 2031 growing at a CAGR of 6.9%”
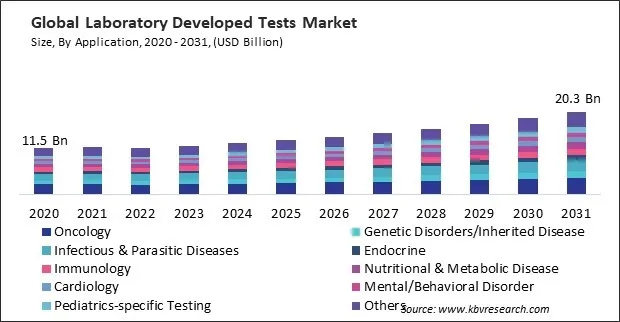
The Global Laboratory Developed Tests Market size is expected to reach $20.3 billion by 2031, rising at a market growth of 6.9% CAGR during the forecast period.
LDTs encompass a wide range of genetic tests designed to diagnose various types of inherited diseases, including single-gene disorders, chromosomal abnormalities, mitochondrial disorders, and complex genetic conditions. Consequently, the genetic disorders/ inherited disease segment would acquire nearly 10% of the total market share by 2031. These tests can identify disease-causing mutations in genes associated with conditions such as cystic fibrosis, muscular dystrophy, inherited cancers, and rare genetic syndromes.
As healthcare expenditure rises, there is often a corresponding increase in demand for advanced diagnostic tests. In addition, with greater investment in healthcare, there’s usually more funding available for research and development of new diagnostic technologies. Hence, increasing healthcare expenditure is driving the growth of the market. Additionally, POCT enables rapid testing and decision-making at or near the point of patient care, leading to quicker diagnosis and treatment initiation. Moreover, POCT facilitates rapid and on-demand testing, which aligns with personalized medicine principles. Hence, the expansion of point-of-care testing drives the market’s growth.
The pandemic disrupted global supply chains, affecting the availability of reagents, consumables, and equipment necessary for LDT development and testing. Laboratories faced challenges in sourcing essential materials, leading to test development and implementation delays. However, the pandemic accelerated the development and deployment of COVID-19 tests, including LDTs, to meet the urgent need for diagnostic testing. Thus, the COVID-19 pandemic had a moderate impact on the market.
However, Multimodal testing often involves integrating multiple testing modalities, such as genomics, proteomics, metabolomics, and imaging, to provide comprehensive diagnostic information. Furthermore, multimodal testing generates large volumes of heterogeneous data from multiple sources, including genomic sequences, protein expression profiles, metabolic pathways, and imaging scans. Therefore, the complexity of multimodal testing is hampering the market’s growth.

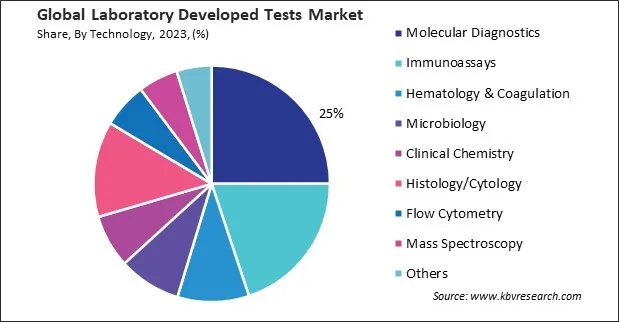
Based on technology, the market is divided into immunoassays, hematology & coagulation, molecular diagnostics, microbiology, clinical chemistry, histology/cytology, flow cytometry, mass spectroscopy, and others. The immunoassays segment attained 19.9% revenue share in the market in 2023. Immunoassays offer high sensitivity and specificity, allowing for accurate detection and quantification of biomarkers even at low concentrations. This capability is crucial for diagnosing conditions early, monitoring disease progression, evaluating treatment efficacy, and detecting relapse or recurrence.
On the basis of application, the market is segmented into oncology, genetic disorders/inherited disease, infectious & parasitic diseases, immunology, endocrine, nutritional & metabolic disease, cardiology, mental/behavioral disorder, pediatrics-specific testing, and others. The infectious & parasitic diseases segment attained a 14% revenue share in the market in 2023. LDTs for infectious and parasitic diseases encompass a wide range of tests designed to detect and identify various pathogens, including bacteria, viruses, fungi, and parasites.
Free Valuable Insights: Global Laboratory Developed Tests Market size to reach USD 20.3 Billion by 2031
Region-wise, the market is analyzed across North America, Europe, Asia Pacific, and LAMEA. In 2023, the North America region witnessed 38% revenue share in the market. The region is a global leader in technological innovation, particularly in the fields of molecular diagnostics, genomics, and personalized medicine. Technological advancements enable the development of sophisticated LDTs that utilize next-generation sequencing, PCR-based assays, microarray technologies, and digital pathology systems.
| Report Attribute | Details |
|---|---|
| Market size value in 2023 | USD 12 Billion |
| Market size forecast in 2031 | USD 20.3 Billion |
| Base Year | 2023 |
| Historical Period | 2020 to 2022 |
| Forecast Period | 2024 to 2031 |
| Revenue Growth Rate | CAGR of 6.9% from 2024 to 2031 |
| Number of Pages | 282 |
| Number of Tables | 390 |
| Report coverage | Market Trends, Revenue Estimation and Forecast, Segmentation Analysis, Regional and Country Breakdown, Porter’s 5 Forces Analysis, Company Profiling, Companies Strategic Developments, SWOT Analysis, Winning Imperatives |
| Segments covered | Technology, Application, Region |
| Country scope |
|
| Companies Included | Neogenomics, Inc., Guardant Health, Inc., Qiagen N.V, Quest Diagnostics Incorporated, Abbott Laboratories, Siemens Healthineers AG (Siemens AG), Illumina, Inc., Bio-Rad Laboratories, Inc., F. Hoffmann-La Roche Ltd. and Eurofins Scientific SE |
By Application
By Technology
By Geography
This Market size is expected to reach $20.3 billion by 2031.
Increasing healthcare expenditure are driving the Market in coming years, however, Complexity of multimodal testing restraints the growth of the Market.
Neogenomics, Inc., Guardant Health, Inc., Qiagen N.V, Quest Diagnostics Incorporated, Abbott Laboratories, Siemens Healthineers AG (Siemens AG), Illumina, Inc., Bio-Rad Laboratories, Inc., F. Hoffmann-La Roche Ltd. and Eurofins Scientific SE
The expected CAGR of this Market is 6.9% from 2024 to 2031.
The Oncology segment led the Market by Application in 2023; thereby, achieving a market value of $4.1 billion by 2031.
The North America region dominated the Market by Region in 2023, and would continue to be a dominant market till 2031; thereby, achieving a market value of $7.5 billion by 2031.
Our team of dedicated experts can provide you with attractive expansion opportunities for your business.

 Drivers
Drivers
 Restraints
Restraints
 Opportunities
Opportunities
 Challenges
Challenges
