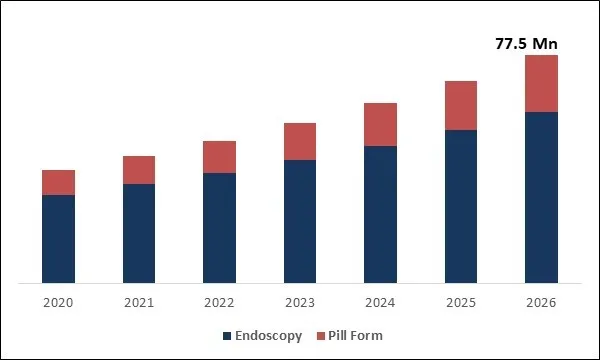
The Global Intragastric Balloon Market size is expected to reach $77.5 Million by 2026, rising at a market growth of 12.4% CAGR during the forecast period. Intragastric balloons are gastric balloons made of soft biocompatible silicone material which is usually filled with sterile saline solutions or gas. These are implanted into non-surgical endoscopic procedures inside the stomach. When inflated, an intragastric balloon will reduce the volume of the stomach which makes a person feel fuller faster and longer. It also helps to minimize food consumption.
Global Intragastric Balloon Market Size
This procedure is an alternative for overweight and obese individuals with an index of body mass (BMI) ranging from 30 to 70 who are not eligible for weight-loss operations. Permanent healthy diet and exercise changes are necessary to ensure the intragastric balloon procedure is a success. The main advantage of this technique is that it helps avoid the complications associated with surgical procedures. Nevertheless, few disadvantages are that not all patients tolerate it, and the possibility of nausea and vomiting. These balloons can also result in weight loss failure if a patient does not adhere to diet and exercise.
A growing trend found on the market is the approval for this medical device by regulatory bodies such as the FDA in the US and its associated treatment followed by the American Society for Metabolic and Bariatric Surgery's acceptance nod. These approvals will likely also be replicated in other countries which will be a positive factor for the Intragastric Balloons industry as their application becomes more standardized.
Intragastric Balloon Market Share
Intragastric balloons have been used progressively in the treatment of obesity and to induce weight loss. Numerous upgrades and technological advances were made in order to achieve optimum results and to reduce the associated complications. Various intragastric balloons have been approved for sale in Europe and South America. Vendors' growing concentration on technical developments, which is one of the key intragastric balloon industry trends, will also drive market growth. Different leading vendors concentrate on improving the capabilities and performance of intragastric balloon devices. The manufacturers also focus on balloon volume changes and longer implantation time.
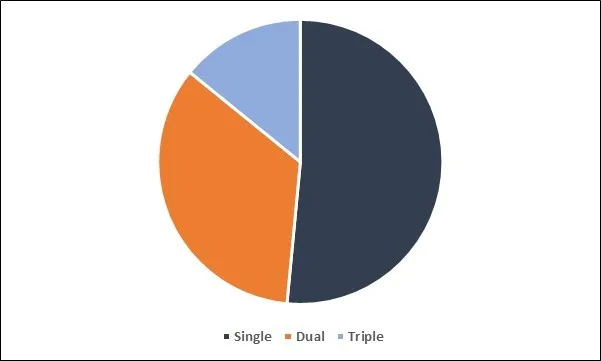
Based on Administration, the market is segmented into Endoscopy and Pill Form. Based on Type, the market is segmented into Single, Dual and Triple. Based on Filling Material, the market is segmented into Gas Filled and saline Filled. Based on End Use, the market is segmented into Clinics, Hospitals and Ambulatory Surgical Centers. Based on Regions, the market is segmented into North America, Europe, Asia Pacific, and Latin America, Middle East & Africa.
Free Valuable Insights: Global Intragastric Balloon Market to reach a market size of $77.5 million by 2026
The market research report covers the analysis of key stake holders of the market. Key companies profiled in the report include Sientra, Inc. (Silimed, Inc.), Apollo Endosurgery, Inc., Obalon Therapeutics, Inc., ReShape Lifesciences Inc., Allurion Technologies, Inc., Helioscopie SA (Sante Actions Group), Districlass Medical SA, Spatz FGIA, Inc., Duomed Group (Life Partners Europe), Medsil.
Market Segmentation:
By Administration
By Type
By Filling Material
By End Use
By Geography
Companies Profiled

The intragastric balloon market is projected to reach USD 77.5 million by 2026.
The major factors that are anticipated to drive the intragastric balloon industry include the increasing global prevalence of obesity, Growing preference of patients towards non-surgical treatment for addressing obesity, and rising demand for Triple Gastric Balloons Demand.
The Hospital segment captured the highest market share in 2019.
Sientra, Inc. (Silimed, Inc.), Apollo Endosurgery, Inc., Obalon Therapeutics, Inc., ReShape Lifesciences Inc., Allurion Technologies, Inc., Helioscopie SA (Sante Actions Group), Districlass Medical SA, Spatz FGIA, Inc., Duomed Group (Life Partners Europe), Medsil.
Our team of dedicated experts can provide you with attractive expansion opportunities for your business.

